The peak of COVID-19 infections might be behind us – at least until the next "variant of concern" of the SARS-CoV-2 virus appears – but the scourge of long COVID lives on. And on.
As defined by the CDC, "post-COVID conditions" encompass a wide range of new, returning, or ongoing symptoms or health problems that people experience at least four weeks following an acute COVID infection. They can include severe fatigue that worsens with exertion, fever, or various respiratory, cardiac, neurological, musculoskeletal, or digestive system symptoms.
Long COVID's manifestations can be serious, even fatal. Last year, Dr. Ziyad Al-Aly and his colleagues described major adverse cardiovascular outcomes at one year following COVID infections. The study exploited a huge U.S. Department of Veteran Affairs database with over five million subjects. It found that beyond the first 30 days after infection, compared to controls, patients with COVID are at increased risk of "cardiovascular disease spanning several categories, including cerebrovascular disorders, dysrhythmias, ischemic and non-ischemic heart disease, pericarditis, myocarditis, heart failure and thromboembolic disease," including a 1.7-fold risk of heart attack and 1.6-fold increased risk of stroke.
They emphasized that "[c]are pathways of those surviving the acute episode of COVID-19 should include attention to cardiovascular health and disease," which, of course, will place significant incremental burdens on the nation's healthcare system going forward. It is estimated that over a million Americans suffer from long COVID, with the numbers continuing to grow. According to the Kaiser Family Foundation, among people with long COVID, 79% report having limitations to their day-to-day activities and 27% characterize the limitations as significant.
A report published last year by a group of Chinese investigators came to conclusions similar to the VA study: "The 12-month risk of incidental cardiovascular diseases is substantially higher in the COVID-19 survivors than the non-COVID-19 controls." The COVID survivors had increased risks of cerebrovascular diseases such as stroke; arrhythmia-related disorders, including atrial fibrillation; inflammatory heart disease such as myocarditis; ischemic heart disease; other cardiac disorders, including heart failure; and thromboembolic disorders such as pulmonary embolism. They had a 1.6-fold increased risk of stroke and double the risk of acute coronary disease. The data offer a useful complement to the Veterans Affairs cohort because the population in this study was much younger, with a mean age of 44.
Finally, a study published in March of gastrointestinal manifestations of long COVID offered additional data. It found that "beyond the first 30 days of infection, people with COVID-19 exhibited increased risks and 1-year burdens of incident gastrointestinal disorders spanning several disease categories including motility disorders, acid related disorders (dyspepsia, gastroesophageal reflux disease, peptic ulcer disease), functional intestinal disorders, acute pancreatitis, hepatic and biliary disease."
The burden of long COVID in addition to the million-plus deaths caused by COVID in the U.S. illustrates the folly of proposals such as the pernicious Great Barrington Declaration, which pushed the fringe notion of "focused protection" of the most vulnerable, while society would forego measures to prevent infection of others.
Supposedly, that would have achieved "herd immunity," but such proposals downplayed the huge death toll that would have resulted, the burden of long COVID even in the "less vulnerable," and the appearance of new virus variants that could escape the immune protection afforded by infection with previous variants. (Note, too, that this approach would have permitted vastly more cases of COVID, each of which would have been marked by in vivo viral replication, more mutations, and new opportunities for Darwinian evolution to select for "fitter" variants.)
The post-infection mechanism of long COVID remains unknown, in spite of intensive study during the pandemic; clinicaltrials.gov lists almost 400 clinical trials related to it.
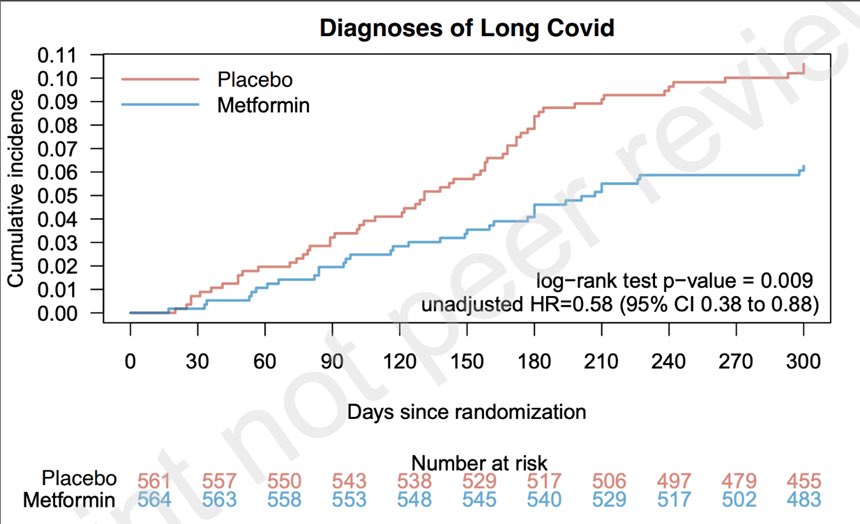
There is one bit of good news. A clinical trial (COVID-OUT) of more than 1,100 randomized participants described in a preprint (neither peer-reviewed nor published yet) found that two weeks of administration of metformin, a drug commonly prescribed for diabetes and pre-diabetes, significantly lowers the risk of long COVID compared to placebo. (The other two drugs in the study, ivermectin and fluvoxamine, had no effect.)
The study included patients ages 30-85 who were overweight or obese, had documentation of a confirmed COVID-19 infection, fewer than seven days of symptoms, no known prior infection, and began drug (or placebo) within three days of their positive test. The study included monthly follow-up for 300 days.
Outpatient treatment with metformin at the time of SARS-CoV-2 infection decreased the development of Long COVID by 42% in this Phase 3 randomized trial, and by over 50% when started less than 4 days from symptom onset. This finding is consistent with the 42% reduction in healthcare utilization for severe Covid-19 with metformin in the first 14 days of the trial.
In other words, two weeks of metformin had both short- and long-term beneficial effects. The effect of the drug on long COVID is shown nicely here:
Dr. Eric Topol, the director of the Scripps Research Translational Institute, has called these findings a "breakthrough," and I agree. Metformin is inexpensive and has a long track record of widespread and long-term use, so two weeks of administration to prevent long COVID should be extremely safe. Inasmuch as nothing else has been shown to lower the risk of long COVID, metformin should be widely prescribed to new COVID patients without delay.
Henry I. Miller, a physician and molecular biologist, is the Glenn Swogger Distinguished Fellow at the American Council on Science and Health. He was the founding director of the FDA's Office of Biotechnology.